This page is intended for UK healthcare professionals only. Prescribing information and adverse event reporting can be found at the bottom of this page.
This promotional content is organised and funded by CarbonNil Medical and is intended for UK Healthcare Professionals.

(potassium citrate/potassium hydrogen carbonate)
8 mEq and 24 mEq prolonged-release granules
Sibnayal® is indicated for the treatment of distal Renal Tubular Acidosis (dRTA) in adults, adolescents and children aged one year and older1
The only alkalizing drug approved for dRTA in Europe and UK2,3
The only sustained-release alkali drug for the treatment of distal renal tubular acidosis2,3
Sibnayal®: A fixed dose combination of 2 alkalizing salts4
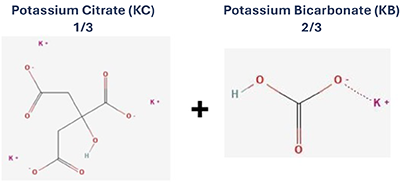
Bicarbonate and Citrate
Provides alkalizing effect necessary to buffer the metabolic acidosis
Citrate
Corrects hypocitraturia. Also acts as a calcium chelating agent reducing calcium salt crystallisation which could lead to nephrocalcinosis and nephrolithiasis
Potassium
Corrects hypokalaemia to help avoid muscle weakness and cardiac disorders
Sibnayal®: An innovative patented formulation4
Granules
- Suitable for achieving prolonged-release oral formulation
- Two-millimeters diameter, favourable size for acceptance in young children/infants
- Two colours: green granules (potassium citrate) and white granules (potassium bicarbonate)
Coating
- With a polymer coating offering a versatile diffusion barrier. Coating thickness appropriately chosen
- Ensures taste masking and good palatability
Prolonged release
- Straightforward option to reduce the number of intakes and maintain activity for an extended period (day & night)
- Provides good gastro-intestinal tolerability by limiting the alkaline load in the stomach
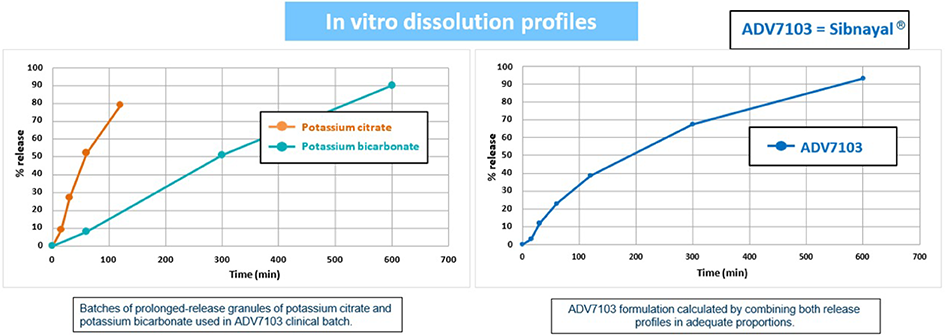
Potassium Citrate is absorbed from the upper part of the intestinal tract while Potassium Bicarbonate is absorbed throughout the digestive tract
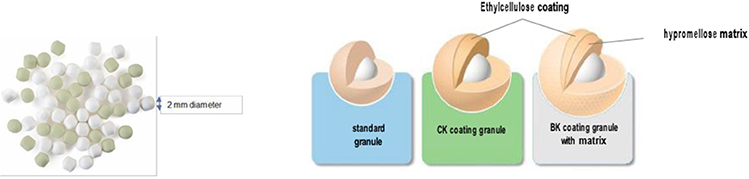
Sibnayal®: An innovative patented formulation, allowing a twice a day intake4
ARENA-1: EU Clinical Development Program
Phase I:B03CS (Guittet C et al. 2020.Scientific Reports.10:13960)
Phase II/III: B21CS-dRTA (Bertholet-Thomas A et al.2021)
Phase III: B22CS-dRTA (Bertholet-Thomas A et al. Pediatric Nephrology.36:1765)
B21CS5
Bertholet-Thomas A et al.2021. Pediatric Nephrology.36:83-91
- Design: multicentre, open-label, non-inferiority, sequential, phase 2/3 study to compare ADV7103 to SoC in dRTA patients
- Aim of the study: to evaluate short-term efficacy, acceptability, tolerability, and safety of ADV7103 in comparison to SoC in adult and paediatric dRTA patients
- Inclusion criteria: males or females, aged 6 months-55 years old, with acquired or inherited dRTA, treated with SoC
- Exclusion criteria: additional proximal tubular signs, hyperkalaemia (plasma potassium >5.0 mmol/L, moderate or severe kidney impairment (GFR <45mL/min/1.73m2), patients receiving potassium-sparing diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, or tacrolimus
- Primary objective: efficacy on metabolic acidosis measured by bicarbonatemia
- Secondary objectives: efficacy on hypokelemia and renal markers, palatability and acceptability, safety and tolerability
- 37 dRTA patient enrolled in 13 centres across France, Serbia and Slovakia
- Inherited dRTA= 36
- Acquired dRTA (Sjögren syndrome) =1
B21CS - Clinical Study Results5,6
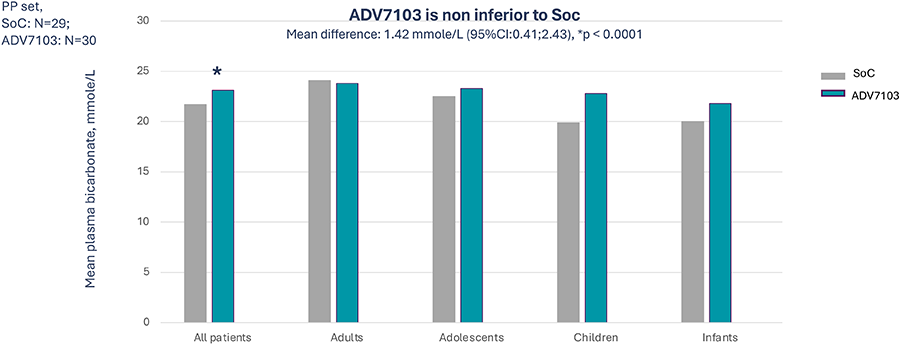
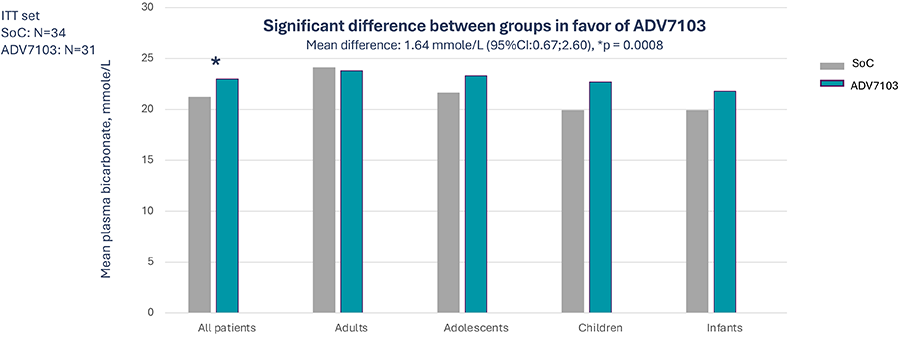
Primary endpoint: Mean pre-morning dose level of plasma bicarbonate over 3 consecutive days (D2 to D4) at alkali steady state (Period 1 vs Period 3)
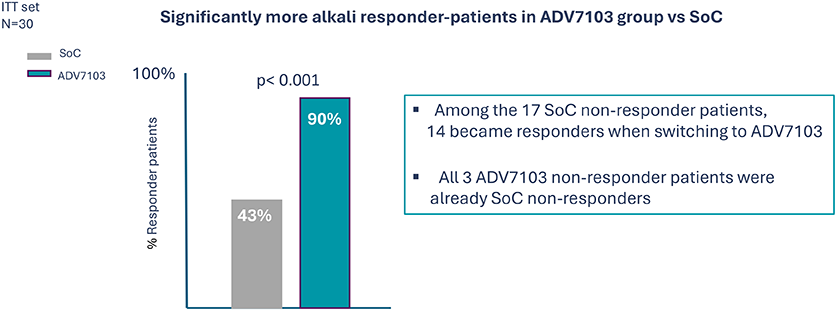
Secondary endpoint: % alkali responder for mean bicarbonate values (all bicarb normal values) over 3 consecutive days (D2 to D4) at alkali steady state (Period 1 vs Period 3)
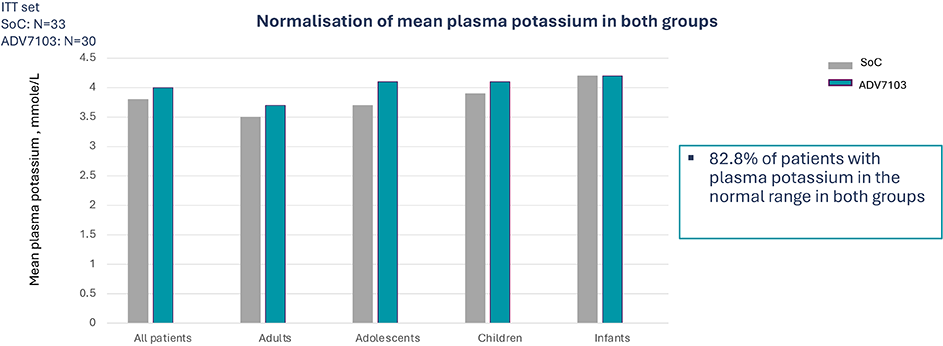
Secondary endpoint: Mean pre-morning dose level of plasma potassium over 3 consecutive days (D2 to D4) at alkali steady state (Period 1 vs Period 3)
Secondary endpoint: Number of alkali treatment daily intakes (Period 1 vs Period 3)

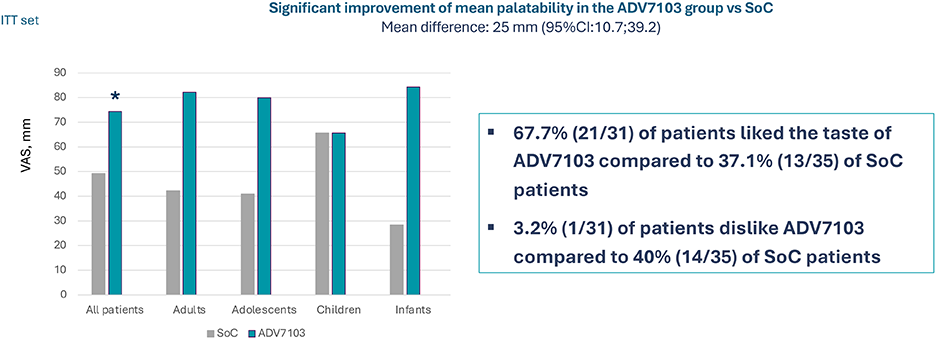
Secondary endpoint: Acceptability / Alkali palatability, VAS from 0 (dislike very much) to 100 (like very much), Period 1 vs Period 3
Secondary endpoint: Tolerability Profile (1)
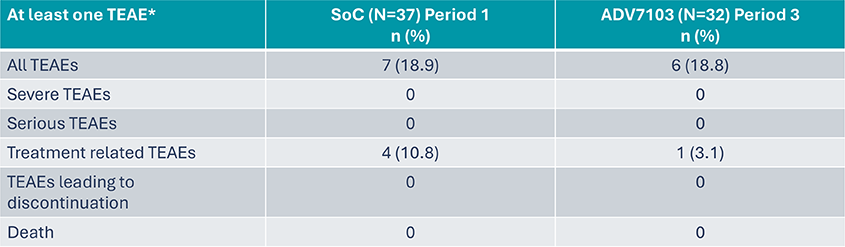
*TEAE: Treatment Emergent Adverse Event
Secondary endpoint: Tolerability Profile (2)
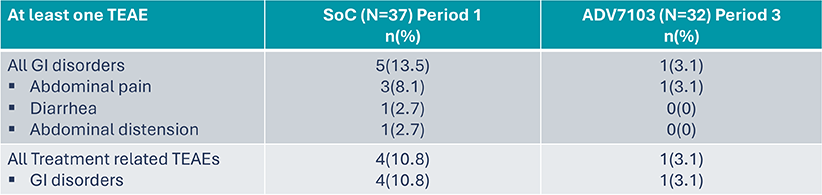
*TEAE: Treatment Emergent Adverse Event
Secondary endpoint: Gastro-intestinal discomfort, VAS from 0 (no complaint) to 100 (extremely severe complaint), Period 1 vs Period 3
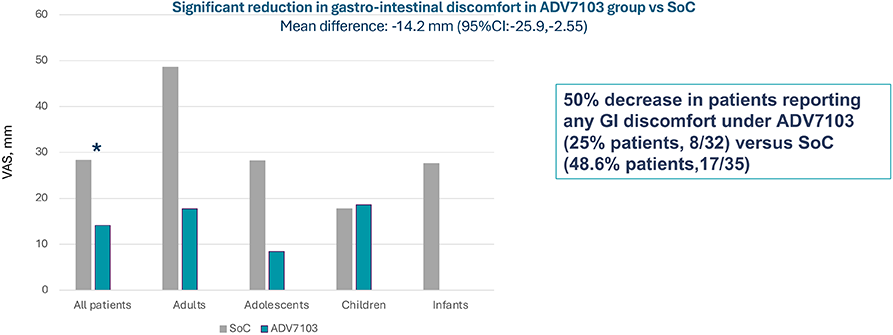
B21CS - Conclusions5,6
- Sibnayal, the first prolonged-release combination of potassium citrate and potassium bicarbonate was demonstrated to be non-inferior to SoC
- As compared to Soc Sibnayal, significantly improves mean plasma bicarbonate levels and improves alkali response rate while maintaining adequate potassium plasma level in patients with dRTA
- Sibnayal allows a twice a day administration, possibly facilitating adherence to alkali treatment, together with its improved palatability in comparison to SoC
- Sibnayal has an acceptable tolerability profile and is significantly better tolerated at a gastro-intestinal level compared to SoC
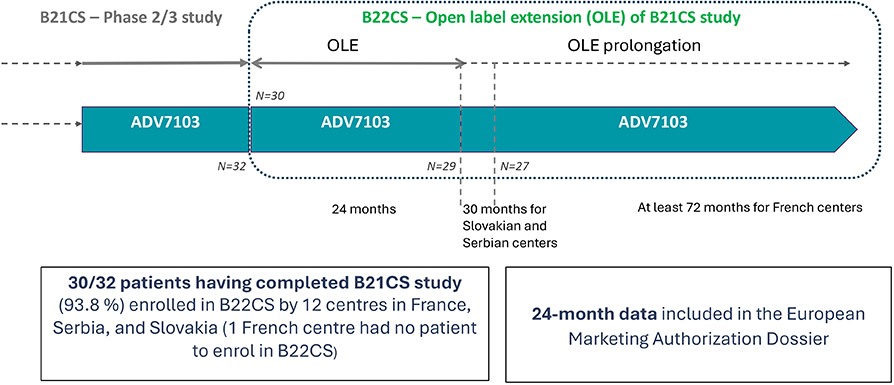
B22CS - Extension Study7,8
- Design: Multicenter, single arm, open-label, extension or follow-up of B21CS study
- Aim of the study: To assess safety, tolerability, and efficacy of ADV7103, and to evaluate adherence. Other clinical outcomes investigated: nephrocalcinosis, nephrolithiasis, estimated glomerular filtration rate (eGFR), bone remodelling, rickets, osteomalacia, growth for children, treatment acceptability, quality of life
- Inclusion criteria: dRTA patients who participated and completed previous B21CS study
- Exclusion criteria: additional proximal tubular signs, hyperkalemia (plasma potassium > 5.0 mmol/L), moderate or severe kidney impairment (GFR < 45 mL/min/1.73 m2), patients receiving potassium-sparing diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, or tacrolimus
- Primary endpoint: long-term safety & tolerability
- Secondary / Exploratory endpoints: Anthropometric and pubertal evaluations, tubular damages and kidney function, bone metabolism, treatment acceptability, QoL
B22CS - Clinical Study Design7,8
B22CS - Results7,8
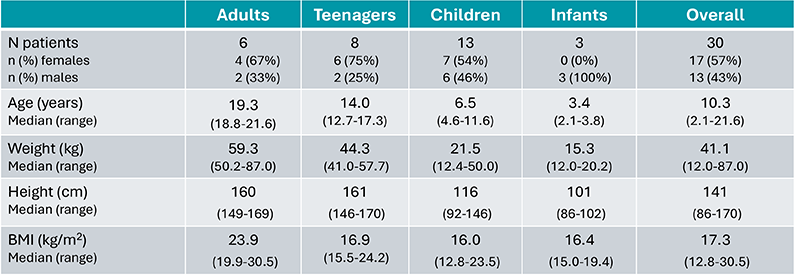
30 patients included: 6 adults, 8 adolescents, 13 children and 3 infants
- 27 patients with data collected above M30
- Baseline mean age±SD:10.6 ±6.0 (min-max:2.0-21.0) years
- EoF* mean age±SD:16.4 ±6.0 (min-max:7.8-28.0) years
All patients with inherited dRTA
- Genetic testing in 22 patients: 21 patients with dRTA type 1 pathogenic variants (1 patient with non identified mutation)
- 9 patients with ATP6V0A4
- 12 patients with ATP6V1B1
- Hearing impairment in 20 patients (all 6 adults concerned)
Mean duration of follow-up in the study: 5.8±1.3 years
*End of Follow-up : last follow-up data available in B22CS
B22CS - Patients Demography7
B22CS – Tolerability profile over the study duration8
Primary endpoint: Number/percentage of patients presenting at least one TEAE*
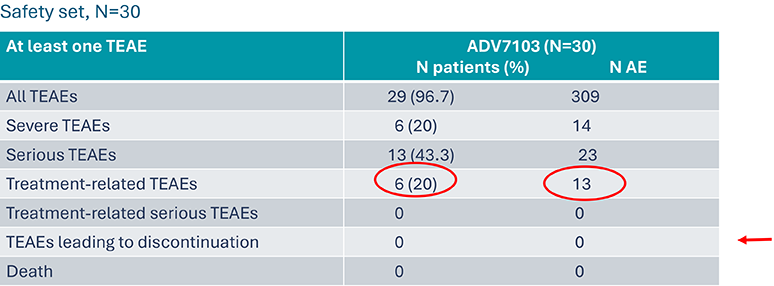
13 related TEAEs and resolved without treatment discontinuation. They concerned 6 patients (20%):
- 11 were gastro-intestinal
- 3 diarrhea in one patient
- 2 abdominal pain in two children
- 2 dyspepsia
- 2 upper abdominal pain
- 1 gastro-intestinal disorder in one adolescent (required dose adjustment with no consequence on plasma and urine metabolic parameters)
- 1 gastro-intestinal pain
- 1 hypokalemia (1 patient)
- 1 nephrolithiasis (1 patient)
No hyperkalemia
*TEAE: Treatment Emergent Adverse Event
B22CS – Compliance over the study duration8
Secondary endpoint: Treatment compliance (according to retrieved study drug), % pts
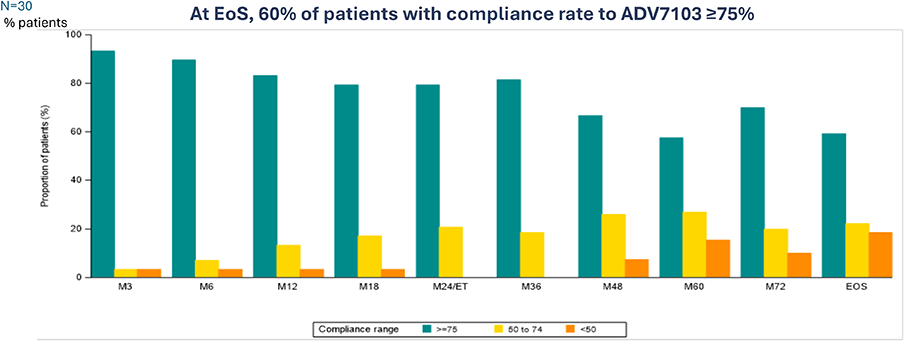
*End of Study: last study visit of B22CS
B22CS – Maintenance of bicarbonatemia over the study8
Secondary endpoint: Mean plasma bicarbonate level
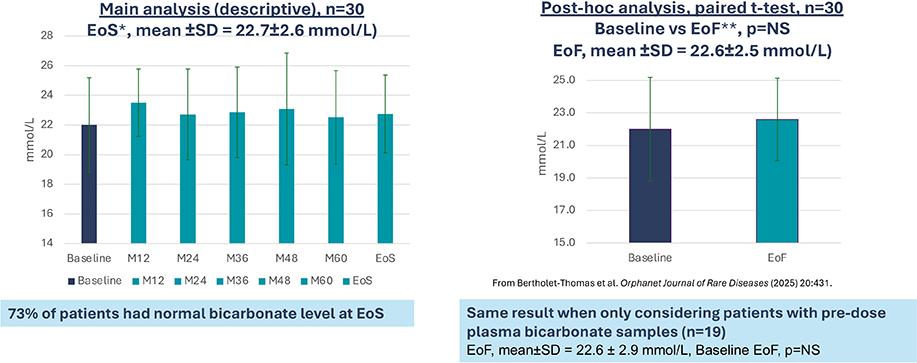
*End of Study: last study visit of B22CS
**End of Follow-up : last follow-up data available in B22CS
B22CS – Maintenance of kalemia over the study8
Secondary endpoint: Mean plasma potassium level
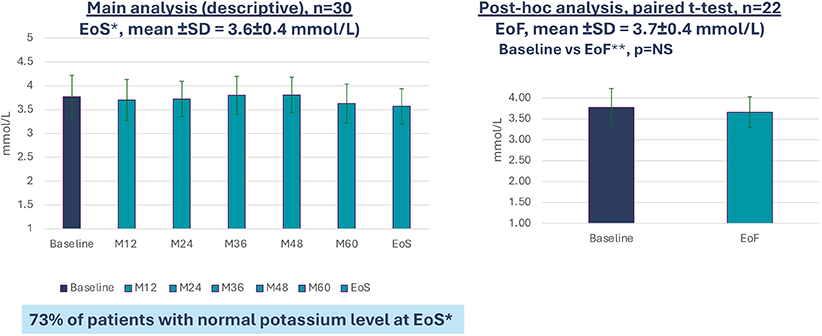
*End of Study
**EoF : End of Follow-up : last follow-up data available in B22CS.
B22CS – Maintenance of eGFR8
Secondary endpoint: Mean eGFR (CKiDU-25) and KDIGO CKD stages breakdown
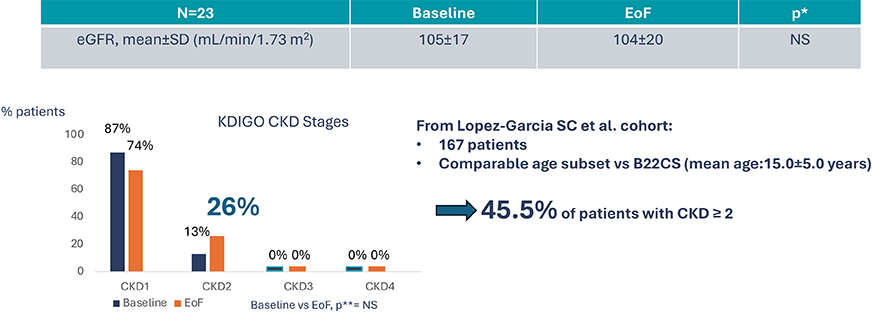
*Post-hoc analysis, paired t-test
**Post hoc analysis, Mc Nemar test
B22CS – Significant increase in height without change in BMI8
Exploratory criteria: Height
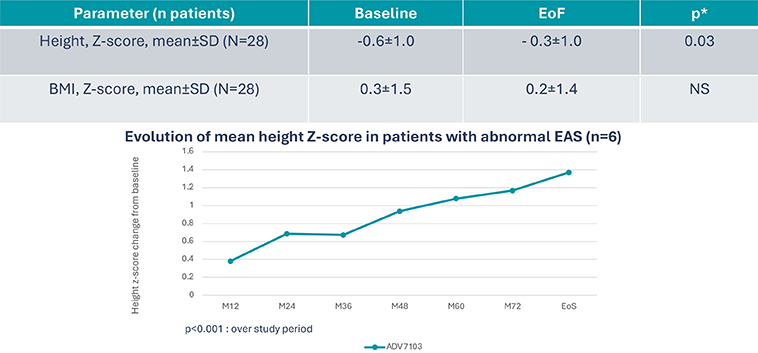
*Post hoc analysis, paired t-test
B22CS – No change in urinary ratios, crystalluria, nephrolithiasis and nephrocalcinosis8
Exploratory endpoints: urinary ratios, crystalluria, nephrolithiasis, nephrocalcinosis
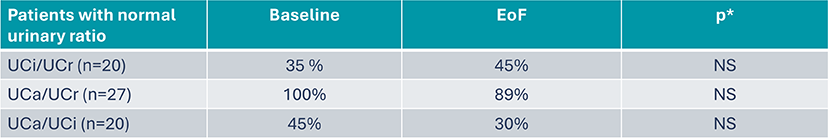
- % patients with positive crystalluria tended to decrease: 53% at baseline vs 35% at EoF, p*=NS
- 70% of patients with UpH between 7-8 (Baseline and EoF)
- No impact on ACCP crystals presence when UpH was > 8 (ACCP: 47% of patients at inclusion versus 35% at EoF, p*=NS)
- Nephrolithiasis: 21% of patients at Baseline vs 44% at EoS
- Nephrocalcinosis: 86% of patients at Baseline vs 92% at EoS
*Post hoc analysis, mac Nemar’s test
ACCP: Amorphous Carbonated Calcium Phosphate
B22CS – Bone metabolism parameters evolution8
Exploratory endpoints: Bone metabolism parameters
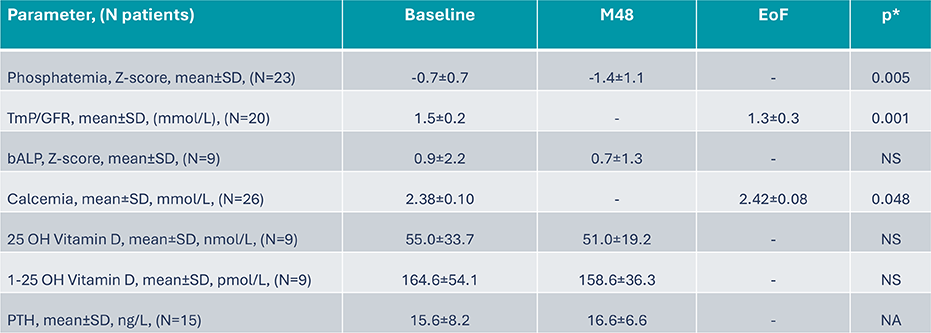
*Post hoc analysis, paired t-test
B22CS – Significant increase in lumbar Bone Mineral Density8
Exploratory endpoints: Lumbar Bone Mineral Density
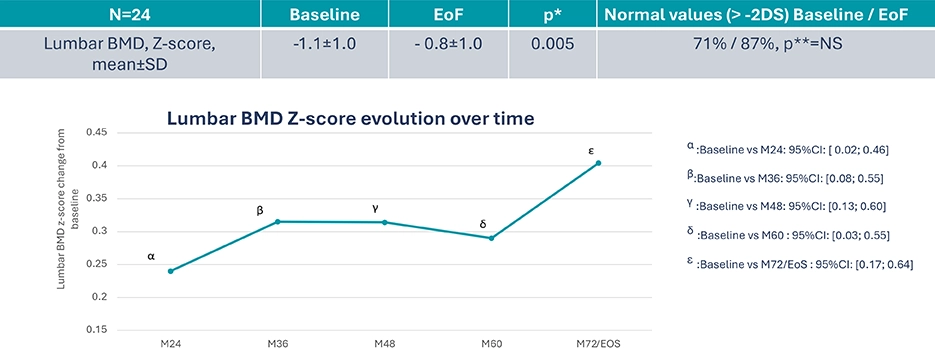
*Post hoc analysis, paired t-test
**Post hoc analysis, Mac Nemar’s test
B22CS – Significant increase in lumbar BMD in pre and post pubertal patients8
Exploratory endpoints: Lumbar BMD in pre and post pubertal patients
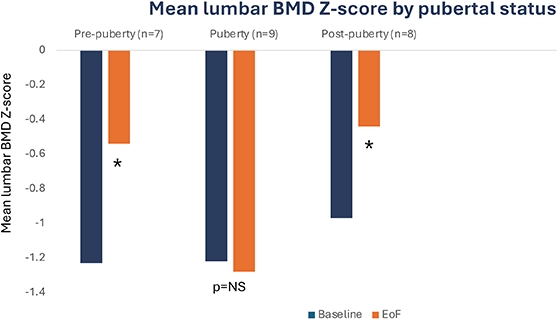
;
*Post hoc analysis, paired t-test, p<0.05
B22CS - 24-month Results7
Exploratory endpoints: Quality of life (quantitative assessment at M6 and M24) and treatment acceptability about switch from SoC to ADV7103 using a VAS

B22CS – 60-month Results9
Exploratory endpoints: Quality of life (qualitative assessment at M60) following switch from SoC to ADV7103, 1-hour interviews
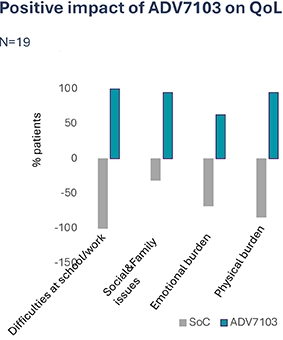
- Mean patients’ satisfaction score with ADV7103: 90%
- Several QoL domains improved when treatment switched from SoC to ADV7103:
- School/work life: e.g. not interrupted/affected by burdensome number of intakes
- Social/family life: e.g. travel/holidays/social activities/family relationship easier, not thinking about managing treatment during day/night
- Emotional functioning: e.g. absence of invasive questions related to the treatment
- Physical health: e.g. reported neutral taste, absence of bad breath, absence of GI disorders, and less strong physical impacts (reported by only 16%, n=3)
B22CS – Conclusions8
- In the long-term, treatment with Sibnayal® allows the control of metabolic acidosis and the prevention of dRTA complications with a good safety and tolerability profile
- Sibnayal® significantly improves growth and lumbar spine BMD in paediatric and young adult dRTA patients
- Most importantly, kidney function is preserved over 6 years on average
- Overall, these results contribute to improving the management and outcomes of this orphan disease
Sibnayal® – Conclusion4,5,7,8
- First and only registered sustained release alkali treatment in the treatment of patients with dRTA
- A simplified posology with only two daily intakes to cover 24h
- A demonstrated efficacy versus SoC in the management of metabolic acidosis with an improved palatability and gastro-intestinal tolerability
- A positive impact on several dimensions of QoL
- A good tolerability profile over the long-term
- A long-term maintenance of efficacy with positive impact on growth, bone mineralization, and kidney function
Sibnayal® composition1,6
- 2 available dosages: 8mEq and 24mEq
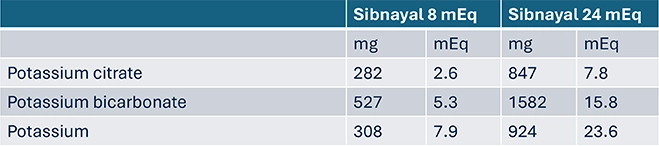
Sibnayal® posology1
- Dose is based on age and weight
- 2 dosages of sachets (stick-packs) available: 8 mEq and 24 mEq, prolonged-release granules
- Maximum dose, regardless of age group is either 10 mEq/kg/day or 336 mEq/day
- The total daily dose should be administered in two intakes. For each individual patients, the dosenearest to the target dose should be fixed by combining whole sachets of the two available dosages
- In case of vomiting within two hours after intake, the patient should take another dose
- The use of this medicine requires medical supervision
Sibnayal® initiation as first alkali1
- When initiating alkalising therapy, the target starting daily dose indicated below for each age group should be used and incrementally titrated to obtain the optimal dose that provides adequate metabolic acidosis control, based on plasma bicarbonate levels
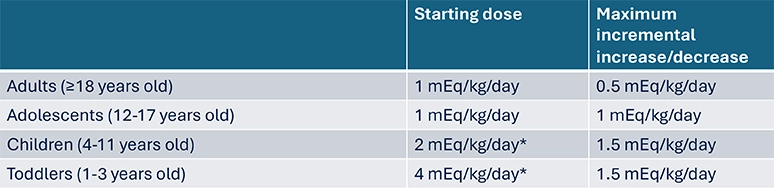
Dose is higher in infants and children due to an increase acid load subsequent to higher metabolic rate and associated higher protein intake, as well as acid generation from skeletal mineralisation6
Switching from SoC to Sibnayal®1
- When switching from another alkalizing therapy, Sibnayal should be initiated at the target dose used with the previous alkalizing therapy (in mEq/kg/day) and titrated where necessary
- Dosage of Sibnayal sachets in mEq corresponds to their content in alkali, so bicarbonate plus citrate, consequently, the potassium content in mEq is the same as that of bicarbonate plus citrate
- Bicarbonate is a monovalent anion so its dose in mEq is the same as in mmole
- Citrate is a trivalent anion, so its dose in mEq is 3-fold that in mmole
- Potassium is a monovalent cation so its dose in mEq is the same as in mmole
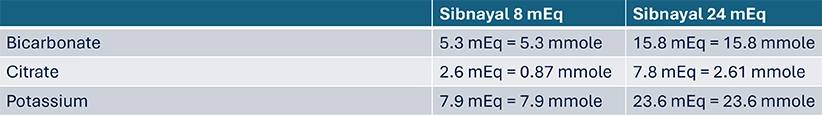
- When switching from another alkalizing therapy, Sibnayal should be initiated at the target dose used with the previous alkalizing therapy (in mEq/kg/day) and titrated where necessary
- SoC dose is to be first converted in mEq as it is often given in mmole
- If SoC is a monovalent anion (e.g bicarbonate), the content in mEq is the same as in mmole
- If SoC is a trivalent anion (e.g citrate), the content in mEq is 3-fold that in mmole
- Then the converted alkali dose of SoC into mEq is to be given in 2 daily intatkes, 1 morning intake and 1 evening intake
- In cases where the SoC does not contain potassium, the potassium given as supplementation must be stopped as Sibnayal provides potassium in addition to alkali treatment
- The maximum dose, regardless of age group, is either 10mEq/kg/day or a total daily dose of 336 mEq, whichever is lower
Sibnayal® administration1
- 2 oral administrations per day, every 12 hours, swallowed with a large glass of water
- The full dose of granules per intake can be swallowed in several smaller portions, if necessary but the content of each sachet must be taken entirely
- Doses should be taken preferably during meals
- For patients who are unable to swallow granules as described above, the granules may be mixed (without crushing) with small amounts of soft food (e.g fruit puree, yoghurt). The soft food mixture must be used immediately and cannot be stored. The mixture should be swallowed without chewing. Care should be taken to ensure that Sibnayal is not retained in the mouth
- In no instance must granules be mixed with hot food, hot liquid or alcohol or chewed or crushed as this can disrupt their prolonged release properties and may lead to a large sudden release of alkalising agent that could affect product efficacy and safety
- Sibnayal granules are not suitable for administration via feeding tubes due to high risk of obstructing the tubes
Sibnayal® special populations1
- Elderly: No dose adjustment is required
- Renal impairment: Sibnayal should only be used in individuals with glomerular filtration rate (GFR) >44mL/min/1.73m2. For individuals with GFR between 45 and 59mL/min/ 1.73m2 Sibnayal should only be used if the potential benefits are considered to outweigh the potential risks
- Hepatic impairment: There is no need for specific target starting daily dose adjustment in patients with hepatic impairment
- Paediatric population: The safety and efficacy of Sibnayal in children below one year of age have not been established. No data are available
Sibnayal® contra-indications1
- Hypersensitivity to the active substance or to any of the excipients
- Renal impairment with GFR ≤ 44mL/min/1.73m2
- Hyperkalaemia
Sibnayal® special warnings and precautions of use1
Hyperkalaemia and cardiotoxicity
Sibnayal® should be used with caution in patients who have conditions pre-disposing them to hyperkalaemia, such as renal impairment, or crush syndrome, as a further rise in plasma potassium may lead to cardiac arrest. Close monitoring of plasma potassium in patients at risk is required at starting dose and after a new dose increase or in the case of worsening of pre-existing disease. Then, the frequency is according to the physician's criteria, but at least twice a year
Sibnayal® should be used in caution in case of combination with other products increasing plasma potassium or predisposing to cardiac dysrhythmia
Potassium content
Sibnayal® 8 mEq contains 308mg of potassium per sachet. This has to be taken into consideration if the patient has a reduced kidney function or if the patient is on a controlled potassium diet
Sibnayal® 24 mEq contains 924mg of potassium per sachet. This has to be taken into consideration if the patient has a reduced kidney function or if the patient is on a controlled potassium diet
Gastrointestinal disorders
Sibnayal® should be used in caution in patients having gastro-intestinal disorders as they could affect efficacy and safety, such as malabsorption, delayed gastric emptying, diarrhoea, nausea, vomiting. In such cases the blood bicarbonate levels should be regularly monitored, and dose adjusted to maintain within normal ranges
The matrix of the granules can be found in the stools, which does not affect the efficacy or safety of Sibnayal®
Renal insufficiency
Sibnayal® should only be used in individuals with glomerular filtration rate (GFR) >44mL/min/1.73m2. For individuals with GFR between 45 and 59mL/min/ 1.73m2 Sibnayal should only be used if the potential benefits are considered to outweigh the potential risks. For these patients doses should be adjusted by regular monitoring of plasma bicarbonate and potassium. Special care should be taken in elderly people in whom renal function can be decreased
Sibnayal® fertility, pregnancy and lactation1
Pregnancy
There are no data from the use of Sibnayal® in pregnant women
Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryonal/foetal development, parturition or postnatal development
Sibnayal® should only be used during pregnancy if the expected benefits outweigh the potential risks. Although during pregnancy and more so during labour, there is more risk associated to a potentially severe acidosis and hypokalaemia in dRTA patients than to alkali treatment, in women with problem pregnancies there might be an increased risk to develop hyperkalaemia when potassium intake is high
Breast-feeding
Potassium is excreted in human milk, but at therapeutic doses of Sibnayal® no effects on the breastfed newborns/infants are anticipated
Sibnaya® can be used during breast-feeding
Fertility
Potassium citrate and potassium hydrogen carbonate are not known to affect fertility
Sibnayal® undesirable effects1
Summary of the safety profile
The most frequently reported adverse reactions are abdominal pain (14%, very common), upper abdominal pain (8%, common) and gastro-intestinal pain (2%, common). Nausea (2 %, common) can be experienced at initiation of therapy
Gastrointestinal disorders description
Very common: abdominal pain (14%)
Common: abdominal pain upper (8%) diarrhoea, dyspepsia, gastrointestinal disorder, gastrointestinal pain (2%), nausea (2%) and vomiting
Gastrointestinal pain, abdominal pain and upper abdominal pain were generally of mild or moderate intensity and resolved within 24 hours without the need to modify or stop treatment. All other gastrointestinal adverse reactions (dyspepsia, vomiting, diarrhoea) were also of mild or moderate intensity, and resolved within 1 to 3 days, without modification or interruption of treatment
Paediatric population
In clinical trails, although numbers were small, the safety profile was comparable in treated patients for adults (N=16 healthy subjects and 7 dTRA patients) and paediatric population (N=27, including 10 adolescents (12-17 years old inclusive), 14 children (4-11 years old inclusive) and 3 infants (6 months-3 years inclusive))
References
- Sibnayal® Summary of Product Characteristics 2023
- European Medicines Agency (EMA) https://www.ema.europa.eu/en/medicines/human/EPAR/sibnayal [Accessed November 2025]
- National Institute for Health and Care Excellence (NICE) https://www.nice.org.uk/guidance/ta838/documents/1#:~:text=action,daily%2C%20typically%20twelve%20hours%20apart [Accessed November 2025]
- Guittet C et al. 2020.Scientific Reports.10:13960
- Bertholet-Thomas A et al.2021. Pediatric Nephrology.36:83-91.
- Epar Public Assessment (EMA /1419 /2021) https://www.ema.europa.eu/en/medicines/human/EPAR/sibnayal [Accessed November 2025]
- Bertholet-Thomas A et al. 2021. Pediatric Nephrology.36:1765-1774
- Bertholet-Thomas et al. Orphanet Journal of Rare Diseases (2025) 20:431
- Acquadro M et al., 2022. Orphanet J of Rare Diseases.17:141.
CLICK HERE for Sibnayal® (potassium citrate/potassium hydrogen carbonate) prescribing information
Adverse event reporting: Reporting suspected adverse reactions after authorisation of the medicinal product is important. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in Google Play or Apple App Store. Adverse events should also be reported to Advicenne@vigipharm.fr. Information about this product, including adverse reactions, precautions, contraindications, and method of administration can be found in the full SmPC. For a copy of the SmPC or further information please contact Advicenne@vigipharm.fr
Ordering
If you are a Healthcare Professional and wish to place an order, please contact us via email; ukenquiries@chapperhealthcare.com
Medical Information requests
If you have a medical information request, please contact us via email; info@carbonnilmedical.co.uk
PP/CNM/2026/019a February 2026

